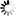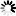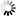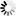PubMed=212198; DOI=10.1016/0092-8674(78)90101-0
Raschke W.C., Baird S., Ralph P., Nakoinz I.
Functional macrophage cell lines transformed by Abelson leukemia virus.
Cell 15:261-267(1978)
PubMed=3792703; DOI=10.1111/j.1432-0436.1986.tb00568.x
Leenen P.J.M., Jansen A.M.A.C., van Ewijk W.
Murine macrophage cell lines can be ordered in a linear differentiation sequence.
Differentiation 32:157-164(1986)
PubMed=16948499; DOI=10.1290/0510075.1
Cuetara B.L.V., Crotti T.N., O'Donoghue A.J., McHugh K.P.
Cloning and characterization of osteoclast precursors from the RAW264.7 cell line.
In Vitro Cell. Dev. Biol. Anim. 42:182-188(2006)
PubMed=19057837; DOI=10.1007/s00774-008-0018-6
Vincent C., Kogawa M., Findlay D.M., Atkins G.J.
The generation of osteoclasts from RAW 264.7 precursors in defined, serum-free conditions.
J. Bone Miner. Metab. 27:114-119(2009)
PubMed=19144319; DOI=10.1016/j.immuni.2008.11.006
Trost M., English L., Lemieux S., Courcelles M., Desjardins M., Thibault P.
The phagosomal proteome in interferon-gamma-activated macrophages.
Immunity 30:143-154(2009)
PubMed=22735334; DOI=10.1038/msb.2012.21
Bordbar A., Mo M.L., Nakayasu E.S., Schrimpe-Rutledge A.C., Kim Y.-M., Metz T.O., Jones M.B., Frank B.C., Smith R.D., Peterson S.N., Hyduke D.R., Adkins J.N., Palsson B.O.
Model-driven multi-omic data analysis elucidates metabolic immunomodulators of macrophage activation.
Mol. Syst. Biol. 8:558-558(2012)
DOI=10.5897/IJBMBR2013.0154
Iloki Assanga S.B., Gil-Salido A.A., Lewis Lujan L.M., Rosas-Durazo A., Acosta-Silva A.L., Rivera-Castaneda E.G., Rubio-Pino J.L.
Cell growth curves for different cell lines and their relationship with biological activities.
Int. J. Biotechnol. Mol. Biol. Res. 4:60-70(2013)
PubMed=23430347; DOI=10.1007/s10616-013-9545-7
Almeida J.L., Hill C.R., Cole K.D.
Mouse cell line authentication.
Cytotechnology 66:133-147(2014)
PubMed=25277546; DOI=10.1186/1471-2164-15-847
Didion J.P., Buus R.J., Naghashfar Z., Threadgill D.W., Morse H.C. III, Pardo-Manuel de Villena F.
SNP array profiling of mouse cell lines identifies their strains of origin and reveals cross-contamination and widespread aneuploidy.
BMC Genomics 15:847.1-847.11(2014)
PubMed=25504905; DOI=10.1002/pmic.201400431
Guo M., Hartlova A.S., Dill B.D., Prescott A.R., Gierlinski M., Trost M.
High-resolution quantitative proteome analysis reveals substantial differences between phagosomes of RAW 264.7 and bone marrow derived macrophages.
Proteomics 15:3169-3174(2015)
PubMed=29889899; DOI=10.1371/journal.pone.0198943
Taciak B., Bialasek M., Braniewska A., Sas Z., Sawicka P., Kiraga L., Rygiel T., Krol M.
Evaluation of phenotypic and functional stability of RAW 264.7 cell line through serial passages.
PLoS ONE 13:E0198943-E0198943(2018)
PubMed=31220119; DOI=10.1371/journal.pone.0218412
Almeida J.L., Dakic A., Kindig K., Kone M., Letham D.L.D., Langdon S., Peat R., Holding-Pillai J., Hall E.M., Ladd M., Shaffer M.D., Berg H., Li J.-L., Wigger G., Lund S., Steffen C.R., Fransway B.B., Geraghty B., Natoli M., Bauer B., Gollin S.M., Lewis D.W., Reid Y.A.
Interlaboratory study to validate a STR profiling method for intraspecies identification of mouse cell lines.
PLoS ONE 14:E0218412-E0218412(2019)
PubMed=31947698; DOI=10.3390/ijms21020538
Mira-Pascual L., Tran A.N., Andersson G., Nareoja T., Lang P.
A sub-clone of RAW264.7-cells form osteoclast-like cells capable of bone resorption faster than parental RAW264.7 through increased de novo expression and nuclear translocation of NFATc1.
Int. J. Mol. Sci. 21:538.1-538.19(2020)
PubMed=33389257; DOI=10.1007/s10096-020-04106-0
Wurtz N., Penant G., Jardot P., Duclos N., La Scola B.
Culture of SARS-CoV-2 in a panel of laboratory cell lines, permissivity, and differences in growth profile.
Eur. J. Clin. Microbiol. Infect. Dis. 40:477-484(2021)



 :2306932016
:2306932016 :021-55150076
:021-55150076